Getting to the bottom of the lithium amount in EV battery is the difference between a whiteboard calculation and current production systems. There are 2 different figures around – about 50 to 60kg per 70kWh battery and 5 to 7kg of elemental Li. Why the differences? This makes a huge impact on the figures used by mining companies of lithium, lithium for Gigafactories and is there enough lithium?
Chemistry Explained
Lithium is expressed in 4 forms.
- Lithium elemental Li with atomic mass of 6.94 u
- Lithium carbonate Li2CO3 or commonly called LCE (99.95% pure lithium carbonate equivalent) and used in lithium nickel ion batteries (with or without cobalt). Li is 18.7% by atomic number. (Molecular weight of Li2CO3 is 73.9)
- Lithium hydroxide LiOH – used in Lithium Phosphate batteries (LiFeP04). Molar mass of LiFeP04 is 157.7 and Li molar mass is 4.3%
- Li2O or Lithium Oxide. Molecular weight of 30, and lithium is 14 of that.
We have to understand that in the cathode of the battery, the only thing important is that lithium is stripped out of the carbonate/hydroxide and it is only Li that provides the energy.
Lithium amount in EV battery by Type
| Measure | Lithium-Ion (Nickel) | Lithium-Ion Phosphate Battery |
|---|---|---|
| Normal Battery Size | 70kWhr | 100kWhr |
| Voltage | 3.7v | 3.2V |
| Energy Density | Li2CO3 = 220mAh/g | LiFeP04 = 160mAh/g |
| Energy Capacity Weight | 816Wh/kg | 512Wh/kg |
| Cathode Weight | 79.6kg | 127kg |
| Lithium as % | 7% | 4.3% |
| Amount of Lithium (kg) | 5.6kg | 5.7kg |
| Theoretical Amount | 30kg | 30kg |
| Current Amount of LCE | 60kg | 130kg |
Know Which Lithium Is Quoted
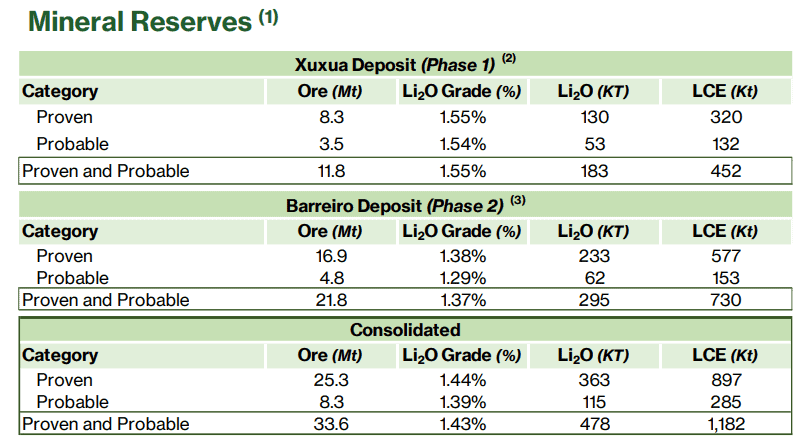
Here is a table from Sigma Minerals on their Lithium Reserves. Take the consolidated figure of proven resources
- 25.3 Million tonnes of ore
- Li2O grade of 1.44%
- Total Li2O =363,000 tonnes (1.44% times 25.3 mt of ore)
- Total LCE = 897,000 tonnes.
- Note that 363,000 times 73.9/25.3 does not make sense so it is unclear whether they are introducing some manufacturing steps or one assumes that it depends on whether it is mined from pegmatite and recovered from the mineral spodumene, and to lesser extent, amblygonite, lepidolite and petalite.
Losses in Production
Efficiencies of the refining process are also less transparent. Lithium is about 1.2% in rocks, concentrated to 6% spodumene (at a 70% recovery) and then refined to 99.95% purity (unknown) for batteries.
Improvement Over Time
There are multiple steps in the process. The use of excess lithium has not been eliminated in NCA or LFP, then there are yield issues (although these are now being recycled back into new batteries.
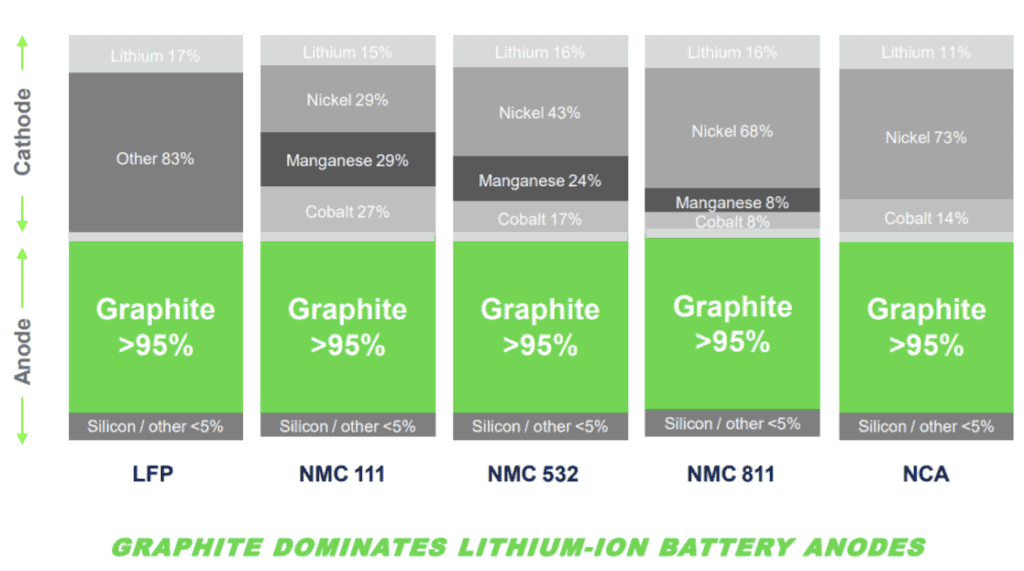
Battery Cathodes and Anode Types
Existing LFP, NMC, and NCA batteries all require graphite, but current graphite prices are about $1200, but lithium has reached over $60,000. Batteries require both cathodes and anodes.